Do I Need To Register With Fda To Get My Label Approved
FAQs
How to use Verification Portal
List of VAT-Exempt Wellness Products pursuant to Republic Human action No. 11534, otherwise known as the "Corporate Recovery and Revenue enhancement Incentives for Enterprises (CREATE) Human action" Effectivity: 02 February 2022
1. What are the products included in the List of VAT-Exempt Health Products?
Under Republic Act (RA) 10963, otherwise known every bit the "Tax Reform for Acceleration and Inclusion or Train Police force", and RA 11534, otherwise known as the "Corporate Recovery and Tax Incentives for Enterprises Act or CREATE Act", the wellness products that are exempted from Value-Added Tax (VAT) are medicines indicated for the prevention and management of diabetes, hypertension, cancer, high cholesterol, mental illnesses, tuberculosis, and kidney diseases, and medicines and medical devices specifically used for the prevention and treatment of COVID-nineteen.
2. Where can I get a copy of the List of VAT-Exempt Health Products?
A copy of the list of medicines for diabetes, hypertension, cancer, high cholesterol, mental illnesses, tuberculosis, and kidney diseases are fastened as Annex A. The list of medicines and medical devices specifically used for the prevention and handling of COVID-19, however, can be accessed at https://doh.gov.ph/VAT-Exempted-Drugs-Listing.
For convenience, a searchable database is uploaded at https://verification.fda.gov.ph, under the tab "VAT-Exempt Health Products".
three. How are medicines included in the List of VAT-Exempt Health Products?
The Department of Wellness (DOH) and the Food and Drug Administration (FDA) place which specific medicines are included in the List of VAT-Exempt Health Products, and this List is transmitted to the Bureau of Internal Revenue (BIR).
The FDA is tasked to update and maintain the list of medicines for diabetes, hypertension, cancer, loftier cholesterol, mental illnesses, tuberculosis, kidney diseases, and COVID-19 medicines and medical devices. To do this, the FDA regularly updates the list based on the list of registered products with the FDA. Products are included in the respective list based on the indication canonical by the FDA.
4. When is the list updated?
Following Articulation DOF-DOH-BIR-FDA Administrative Lodge No. 2-2018 dated 21 December 2018 and Joint DOH-DOF-FDA-BIR-BOC Administrative Club No. 2021-0001 dated 23 June 2021, the FDA must provide an updated list xxx (30) days prior the beginning of every quarter. Thus, the FDA endeavors to publish the list inside the months of February, May, August, and November of every year.
v. How can I request to add, remove, or correct an entry in the Listing?
Requests to add, remove, or correct any entries in the list of medicines for diabetes, hypertension, cancer, high cholesterol, mental illnesses, tuberculosis, or kidney diseases are submitted through the 'Enquiry Form for the List of VAT-Exempt Medicines' at https://bit.ly/vatexemptenquiry. The same form is attainable at the FDA Verification Portal, https://verification.fda.gov.ph. Enquiring entities are requested to complete the following information, in order to appropriately process the query:
a. Generic name
b. Brand name (if applicable)
c. Dosage strength
d. Dosage form and route
e. CPR Number (if available)
f. Indication
g. Blazon of request (inclusion, delisting, correction)
h. Specific listing pertained to in the request (medicines for diabetes, hypertension, cancer, high cholesterol, mental illnesses, tuberculosis, or kidney diseases)
i. Other details of request (e.g. reason for asking, type of correction requested)
Changes to the list are included in the regular quarterly update. Enquiries are consolidated at the end of January, April, July, and October of every yr, to permit sufficient time for the publication of the list for the regular quarterly update.
For changes to the list of medicines and medical devices specifically used for the prevention and treatment of COVID-19, enquiring entities are advised to contact the FDA Policy and Planning Service at [email protected]
6. If the medicine is included equally an entry in the List, but is used for the handling of a disease not identified amongst those under the CREATE Human action, is the medicine notwithstanding considered VAT-Exempt?
The list issued by the FDA intends to identify drugs specifically used for the prevention and management of diseases (i.e. diabetes, hypertension, cancer, high cholesterol, mental illnesses, tuberculosis, and kidney diseases) which have been identified under the TRAIN Police and CREATE Deed. The fields included in the listing accept, thus, been then designed to allow the identification of specific drugs which have been registered with the FDA for the purposes of preventing and treating the identified diseases included in the afore-cited laws.
Nevertheless, it is likewise acknowledged that drug products containing the same agile pharmaceutical ingredients and begetting the same dosage forcefulness and class may remain to be indicated for multiple diseases. In such cases, a stardom can exist made when such drugs are used for the management of a disease other than those identified in the aforecited laws, ergo if a product is not specifically prescribed for the treatment and/or prevention of the identified diseases, information technology should not be construed to be among the medicines which are considered VAT-Exempt pursuant to the TRAIN Law and the CREATE Deed.
Delight be also informed that the FDA is currently undertaking a review of the drug registry database to address this in the long-term, with the aim of improving the accuracy of reflected products in the published listing of VAT-Exempt Products.
7. If the medicine contains the same generic name but is in a dissimilar dosage form, is information technology considered the same as the entry in the List?
No, the fields included in the tabulated list generated by the FDA were so designed in order to identify specific drugs which have been registered with the FDA for the purposes of preventing and treating the identified diseases included in the Train Police and CREATE Act. Thus, if the product bears a dosage form not included in the List, information technology is NOT considered every bit the aforementioned as the entry.
Further note that active pharmaceutical ingredients of such drugs tin can be indicated for multiple diseases, and so in recognition of this, the FDA included the dosage forms of the drug products to accurately produce a list which fits the intention of the provisions of the afore-cited laws. However, every bit noted in FAQ No. 5, there remains to be circumstances which allow for further distinction.
8. Tin can I request for a certification from the FDA that my product is exempted from VAT?
The FDA does not certify if products are considered VAT Exempt. For the purposes of availing VAT Exemption, stakeholders are advised to have reference to the List published by the FDA along with their copy of the Certificate of Product Registration. Should at that place be any discrepancy, entities are advised to submit their queries post-obit FAQ No. 4.
Heart for Drug Regulation and Enquiry
Center for Food Regulation and Inquiry
Complete Guide on How to Apply for a Food CPR Version two
LICENSING OF Food ESTABLISHMENTS
ane. What is a License to Operate (LTO)?
LTO is an authorization issued by the FDA to an establishment to grant permission to undertake a trade or carry out a business concern action, such as manufacturing, importation, exportation, sale, offer for sale, distribution, or transfer of nutrient products.
2. Who will apply for License to Operate?
A sole proprietorship, a partnership, a corporation, an institution, an association, or an organization engaged in whatever of the following activities are being licensed by FDA:
- Manufacturers are establishments engaged in any and all operations involved in the product of health products including training, processing, compounding, formulating, filling, packaging, repacking, altering, ornamenting, finishing and labelling with the cease in view of its storage, sale or distribution.
- Repackers are establishments engaged in the procedure of packaging or changing of container, wrapper (that may include or not a changing of label) from a bulk cloth to retail packaging sizes in furtherance of distribution of food.
- Toll manufacturers are manufacturers that carry contract manufacturing where conditions of the contract are defined, agreed and controlled; and all aspects of contracted work are specified to obtain quality product/s conforming to the agreed standards.
- Benefactor/Importer/Exporter are establishments that that import or export raw materials, ingredients and/or finished products for its own apply or for wholesale distribution to other establishments or outlets. If the distributor/importer/exporter sells to the full general public, it shall be considered a retailer.
- Traders are establishments which are registered owners of food and food products and/or procure the raw materials and packing components, quality control standards and procedures, merely subcontract the manufacture of such product to a licensed manufacturer. In addition, a trader may as well appoint in the distribution and/or marketing of its products.
- Wholesalers are establishments engaged in local distribution of pre-packaged food products in commercial quantity.
3. How to apply for License to Operate using the E-portal System (FOR FOOD MANUFACTURERS)?
East-Portal System
The applicant company must asking for a user account to FDA Action Heart (FDAC) by sending an email to [email protected] with the ff. format:
Discipline: Request for eLTO User Account
BODY:
a) Email address (preferably company e-mail address)
b) Name of the Authorized company representative (preferably permanently employed and not simply a consultant)
c) Position in the company
d) Contact Number
Eastward) Company Name
Then, the bidder company must adhere a notarized Authorization letter for the eLTO User Account Application (please refer to Annex B of FDA Round no. 2016-004 (Procedure on the Utilise of the New Application Form for LTO thru the FDA due east-Portal)
Later on issuance of user business relationship and password, the bidder company can access the east-Portal through https://eportal.fda.gov.ph for filing of awarding
five. How can the applicant company be notified after successfully filing an application?
In the ePortal arrangement, the case application should be under the "PARTICIPATED" binder of the applicant company. If the case application is still in the "DRAFT" folder, the bidder company should go over the case application and click "Continue" to assign the task to payment.
In the eServices organisation, the Application Summary shall be automatically sent to the applicant'southward registered electronic mail address to point the successful submission of the application.
vi. Subsequently filing an application in the system, what is the next step for the applicant visitor?
In the ePortal System, the applicant company should download the Order of payment and pay the prescribed fees indicated in the Lodge of payment.
In the eServices System, the awarding will exist pre-assessed with regards to completeness and correctness of the application vis-à-vis uploaded requirements. Applications with incomplete documentary requirements and/or inconsistent data entries with the submitted documentary requirements based on existing FDA rules and regulations shall not be accustomed and the application will non proceed to the next pace of the process. The applicant company will be notified on the reason/s of disapproval via alleged e-mail. If the application passed the pre-assessment step, the bidder visitor shall receive the Order of Payment with Reference Number through alleged electronic mail indicating the fees to be paid.
. How volition the applicant visitor exist informed on the approval/disapproval of its LTO awarding?
The result of the application shall be sent to the applicant'south registered e-mail.
For eServices Organization, the eLTO will be printed past the applicant company on a standard A4 size (21 cm 10 29.7 cm) newspaper, on full-colored page and in portrait orientation. The printed eLTO shall exist positioned on the most conspicuous place within the business establishments. A QR Code verifier shall be included in the LTO as means of confirmation of the legitimacy of the document.
eight. What are the grounds for disapproval of an LTO application?
The grounds for disapproval of LTO awarding may be any of the following, as stated in AO No. 2020-0017 and AO No. 2014-029;
- Failure to submit articulate, consummate, or compliant documentary requirements;
- Absence of physical office upon inspection, without permission or blessing from FDA;
- The applicant made misrepresentations, false entries, withhold relevant data reverse to the provisions of the law;
- The owner has violated any of the terms and conditions of its license; and
- Such other analogous grounds or causes as determined by the FDA.
The disapproval of an application is without prejudice to re-application. However, disapproval shall mean outright forfeiture of payment.
10. What are the requirements for Initial License to Operate (LTO) application?
The following documents based on AO 2020-017 shall be uploaded using the Due east-Portal. Scanned copy should be 100-150 dpi (dots per inch), maximum of 2MB:
Initial LTO Requirements:
- Application Form (web-based form)
- Amid other information, the bidder shall provide the post-obit information:
- Location Programme
- Global Positioning Organisation (GPS) coordinates
- Proper noun of the Qualified Person
- Proof of Business organisation Name Registration
Whatever ane of the following shall exist submitted equally proof of business name registration (in pdf):
- For unmarried proprietorship, the Certificate of Business organization Registration issued past the Department of Merchandise and Industry (DTI);
- For Corporation, Partnership and other Juridical Person, the Certificate of Registration issued past the Securities and Commutation Commission (SEC) and Articles of Incorporation;
- For Cooperative, the Document of Registration issued past the Cooperative Authority and Articles of Cooperation; or
- For Regime-Owned or Controlled Corporation, the law creating the establishment, if with original charter, or its Certificate of Registration issued by the Securities and Exchange Commission (SEC) and Articles of Incorporation, if without original charter.
- Business organisation Permit- When the business or establishment accost is different from the concern name registration accost, the applicant shall submit a re-create of the Business Let (eastward.grand. Mayor's Allow).
- Proof of Capital or Income such as latest audited Fiscal Statement with Balance Sheet (in pdf) shall be submitted. This is to verify the capitalization of the establishment to their corresponding awarding fees.
- The following documents shall be presented to the FDA inspector for examination or review, when required:
- Risk Management Plan (RMP), which shall be required for medium and large nutrient manufacturers, traders, and distributors (importer, exporter and/or wholesaler)
Site Master File (SMF), which shall exist required for applicants applying for LTO as manufacturers of large and medium food manufacturers (CFRR)
Additional Requirements, as applicable based on FDA Circular 2021-012 (Food Traders/Distributors):
- Franchise agreement– For franchises, a copy of the Franchise understanding is recommended to be submitted. The concern proper name of the establishment reflected in the LTO shall be based on the merchandise name indicated in the franchise understanding. In addition, this must be consequent with the business name registration
- Contract of understanding - For the appropriate determination of activity to be indicated in the LTO, a copy of the Contract of Understanding is recommended to exist submitted. The basis for the LTO activity shall depend on the legally binding contract agreement betwixt the applicant and its client/supplier
xi. What are the requirements for Renewal of License to Operate (LTO) application?
LTO Renewal Requirements:
- Achieved due east-Application Form with Proclamation of Undertaking; and
- Payment of Fees
Annotation: Awarding for renewal shall be washed within iii (3) months prior to the validity date of the LTO. Applications filed after the validity engagement of the LTO shall be subject to surcharge as prescribed in RA 9711 and its IRR.
12. What are the requirements for Major Variation of License to Operate (LTO) awarding?
Requirements for Specific Variation in the LTO
Major Variation for Local Manufacturers
| Blazon of Variation | Requirement |
| Transfer of Location of Manufacturing Plant - Physical transfer of the establishment (and may entail changes in the previously canonical address) | a. Concern let reflecting the new address b. Updated Site Master File to be presented upon inspection |
| Expansion of Manufacturer and/or Boosted Product Line; or Modify of Manufacturing Activity - Expansion shall refer to expansion fabricated which is adjacent to the existing location of the institution - Boosted product line refers to boosted blazon or class of products produced within the aforementioned manufacturing site - Change in manufacturing activeness shall refer to an additional activity that a manufacturer engage in (e.g. LTO every bit Manufacturer with boosted activity as Repacker) | Updated Site Master File to be presented upon inspection |
13. What are the requirements for Minor Variation of License to Operate (LTO) application?
Small Variation
|
14. What is a Qualified Person and the corresponding qualification/credential requirement?
A Qualified Person refers to an organic or full-time employee of the establishment who possess technical competence related to the establishment's activities and wellness products by virtue of his profession, training or experience. A qualified person has the responsibleness to comply with the technical requirements of the FDA or discuss or clarify matters with the FDA when submitting technical requirements or engage the FDA officials when conducting inspection or post-market surveillance activities. The qualified person may likewise be the duly Authorized Person of the institution.
| Qualification | Preparation Requirement |
| Visitor Regulatory Officer (Authorized Person) and Food Safety Compliance Officer who is preferably a graduate of food-related courses including but not limited to food engineering, food and nutrition, chemistry, microbiology, chemical/germ-free applied science, veterinary medicine, fisheries, agronomics (RA 10611) | Certificate of Attendance on seminar on food rubber, GMP or HACCP given by the academe, WHO, FAO, NGOs, cooperative, food industry organizations, professional organizations, or the FDA Academy |
16. What are the references for the acquit of inspection of establishment?
- Republic Deed 9711 (The FDA Act of 2009)
- Republic Act 10611 (The Food Prophylactic Act of 2013)
- Administrative Guild 153 s. 2004 (Revised Guidelines on Electric current Good Manufacturing Practice in Manufacturing, Packing, Repacking or Belongings Food)
- Authoritative Order 2014-0029 Rules and Regulation on the Licensing of Food Establishment
- For iodized salt, Democracy Act 8172
- For bottled water, Administrative Order 18 south. 1993
- For labelling, Administrative Club 2014-0030
17. What are the prescribed fees and charges?
| Nomenclature | Initial (2 years validity) | Renewal (5 years validity) | |
| 1. Food Distributors (Importer, Exporter, Wholesaler) | 8,080.00 | 20,200.00 | |
| 2. Food Manufacturer/Trader | |||
| 2.1 250K and below | i,010.00 | 2,525.00 | |
| 2.ii Over 250K but below 500k | 1,515.00 | three,788.00 | |
| 2.iii 500K merely below 1M | 2,020.00 | 5,050.00 | |
| 2.4 Over 1M just below 5M | iv,040.00 | 10,100.00 | |
| 2.five 5M but below 10M | half dozen,060.00 | 15,150.00 | |
| 2.6 10M but below 20M | x,100.00 | 25,250.00 | |
| 2.7 20M but beneath 50M | xx,200.00 | fifty,500.00 | |
| ii.viii 50M and above | thirty,300.00 | 75,750.00 | |
| 3. Bottled Water Manufacturer/ Trader/ Distributor | 3,030.00 | 5,050.00 (3 years validity) | |
| 4. Iodized Salt Manufacturer/Trader | |||
| 2.1 Large and Medium | 2020.00 | 5,050 | |
| 2.ii Small | 1010.00 | 2,525 | |
| 2.3 Subsistence | 410.00 | 1,010 | |
| five. Iodized Salt Benefactor | 2,020 | 5,050 | |
Note: Surcharges/penalties are imposed for late renewal as per FDA Circular 2011-004.
18. How to pay for an LTO application?
Based on FDA Informational 2021-0246, please refer to the below payment options:
| Payment Option | Credit |
| via Landbank ATM Menu | Existent-time Credit* |
| Other Banks via Bancnet | Next banking Day* |
| Cash Payments via vii/11 and Bayad Eye | Next Banking Day* |
| *for transactions made during holidays and weekends, payment volition be credited on the next banking days | |
Further, FDA is now included in the Landbank Link.BizPortal as biller:
https://www.landbank.com/due east-cyberbanking/other-due east-banking-services/linkbizportal
Applicant companys are too hereby brash that payments through FUND TRANSFER via account number 0392-1030-58 will NO LONGER be accommodated effective immediately since Landbank Link.BizPortal is now available.
20. When will the LTO application exist bailiwick to surcharge and penalization?
An awarding for renewal of an LTO received later on its date of expiration shall be subject to a surcharge or penalty equivalent to twice the renewal licensing fee and an boosted x% per month or a fraction thereof of continuing non-submission of such application up to a maximum of 120 days.
An application for renewal of license filed after 120 days shall be considered expired and the application shall be field of study to a fee equivalent to the full surcharge or penalty plus the initial filing fee and the application shall undergo the initial filing.
cGMP / HACCP CERTIFICATE
ii. How to apply for GMP Document?
Based on FDA Round 2020-026, the FDA Activeness Center (FDAC) receives the letter asking for issuance of GMP/HACCP Document via [email protected],gov.ph . The FDAC personnel will respond to the email and provide to the applicant company the designated document tracking number of the application.
3. What are the requirements for GMP application?
| Requirements | Where to Secure |
| a) Inspection study with document of Compliance/ Recommendation Letter from RFO | FDA Regional Field Office |
| b) Valid LTO | Eye for Food Regulation and Research |
| c) Proof of payment | FDA Cashier/ Other FDA Authorized Payment Portals/ Banks |
5. How long is the validity?
Unless the LTO is revoked, validity of GMP Document shall be coterminous with the LTO validity, whereas the HACCP Certificate shall only be valid for ane (one) year.
DIAMOND SANGKAP PINOY SEAL/ SANGKAP PINOY SEAL CERTIFICATE
ii. How to apply for GMP Certificate?
Based on FDA Round 2020-026, the FDA Activity Center (FDAC) receives the letter asking for issuance of GMP/HACCP Certificate via [email protected] The FDAC personnel will reply to the email and provide to the applicant company the designated document tracking number of the application.
iii. What are the requirements for GMP awarding?
Basic Requirements based on RA 8976 (Food Fortification Law of 2000), RA 8172 (ASIN Police), the Sangkap Pinoy Seal Program Transmission of Operations (Dec 2000) and Authoritative Order No. 82 s. 2003 (Guidelines on the Granting of Diamond Sangkap Pinoy Seal to Manufacturers of Fortified Products):
| Requirements | Where to Secure |
| a) Duly achieved application forms | FDA Website |
| b) Valid LTO | Center for Nutrient Regulation and Research |
| c) Results of product analysis for vitamin A, Iron and Iodine from an FDA recognized laboratory. | Laboratory analysis issued/conducted by FDA-Recognized laboratories. |
| d) Sample label with Sangkap Pinoy Seal/ Diamond Sangkap Pinoy Seal | Applicant Company/ Manufacturer/ Source/ Supplier |
| e) Proof of payment | FDA Cashier/ Other FDA Authorized Payment Portals/ Banks |
| f) Inspection written report with Document of Compliance | FDA Regional Field Office |
four. What are the fees to be paid?
| Dominance | Fees |
| a) Use of the seal (Regular Seal) | P8,000.00 non-refundable fee + i% LRF |
| b) Processing fee for every application (Regular Seal or Diamond Seal) | P500.00 + 1% LRF |
v. How long is the validity?
Unless the LTO is revoked, validity of Diamond Sangkap Pinoy Seal/ Sangkap Pinoy Seal Certificate shall be valid for an indefinite period.
REGISTRATION PRE-Cess
ane. How long does the pre-cess phase commonly take?
For pre-assessment, no specific timeline is committed in the Citizen's Charter. However, our initial target is to process applications for pre-assessment within 7 WORKING DAYS depending on the bulk of applications received per week.
six. Where tin I find/download the result of Pre-assessment?
A organisation-generated electronic mail notification of complete requirements will be sent to the registered E-mail service. On the other mitt, you may also download the result of pre-assessment under the Generated Documents tab in the instance number. Right click on the instance number. Select "Summary" and look for the Generated Documents tab. In one case the document appears, tick the file then click download.
REGISTRATION OF PRE-PACKAGED, Processed FOOD PRODUCTS (ISSUANCE OF CERTIFICATE OF Production REGISTRATION)
1. What are the products that need to secure Document of Product Registration in the Philippines?
All processed food and food products including food additives, food supplements and bottled h2o manufactured and/or distributed (i.eastward. imported, exported and/or wholesale for local distribution), for trade and/or repacked are required to secure a Document of Production Registration earlier these are sold, offered for sale or use, distributed or supplied, among other marketing and promotional activities as per R.A. 9711 and A.O. 2014-0029.
2. Practise I demand to secure a CPR for the Raw Materials that I will import for my own use as manufacturer?
Food establishments with License to Operate equally Manufacturer and Trader who directly import and apply raw materials, ingredients and food additives for their ain use or for further processing to industry a candy food production, need not secure a CPR for the raw materials, ingredients and food additives. However, the sources of these raw materials should be notified to FDA to be reflected in the Licensing Database of Center for Food Regulation and Inquiry.
3. What are the requirements to secure a CPR?
You may refer to FDA Round No. 2020-033 "Procedure for the Use of the Modified Electronic Registration System for Raw Materials and Prepackaged Processed Food Products Repealing FDA Round NO. 2016-014 "Procedure for the Use of Electronic (E-Registration) System for Pre-Packaged Processed Food Products" to include Guidelines for Pre-cess and Reiteration of Pre-Assessment Procedures in Applying for Document of Production Registration for Food prior to filing for registration. For farther guidance on registration requirements and nutrient regulations, please refer to Authoritative Social club No. 2014-0029 (Rules and Regulations on the Licensing of Food Establishments and Registration of Processed Nutrient, and Other Nutrient Products, for Other Purposes). You need to fill upward Initial Application Class and upload a scanned copy of the following documents in the system during application:
- Compliant labels in accordance with the prevailing labelling guidelines stipulated in Administrative Order No. 2014-0030
- Pictures of the product in all angles from at least ii different perspectives assuasive visual recognition of a production as the aforementioned with the 1 being registered pictures of the product in all angles
- Clear and complete loose labels or artworks, as applicative, of all packaging sizes, or equivalents as defined by FDA regulations except for bulk raw materials, ingredients and food additives intended for farther processing or for distribution to establishments/ manufacturers for further processing.
As applicable, documents to substantiate claims, such equally technical, nutritional or health studies or reports, market-research studies, Document of Analysis, quantitative assay and computations, scientific written report or studies published in peer-reviewed scientific journals, certificates or certification to support employ of logo/seal on Sangkap Pinoy, Halal, Organic, or Kosher food and in compliance with electric current labelling regulations.
All applications for food product registration shall be lodged in the Modified E-portal version 2 (https://eportal.fda.gov.ph) and shall undergo pre-assessment (FDA Circular 2020-033-A).
iv. How to secure CPR for nutrient products using E-portal Version 2?
All nutrient establishments applying for product registration should have a valid License to Operate issued by FDA. For locally produced products, the product being practical should be included in the LTO of the manufacturer.
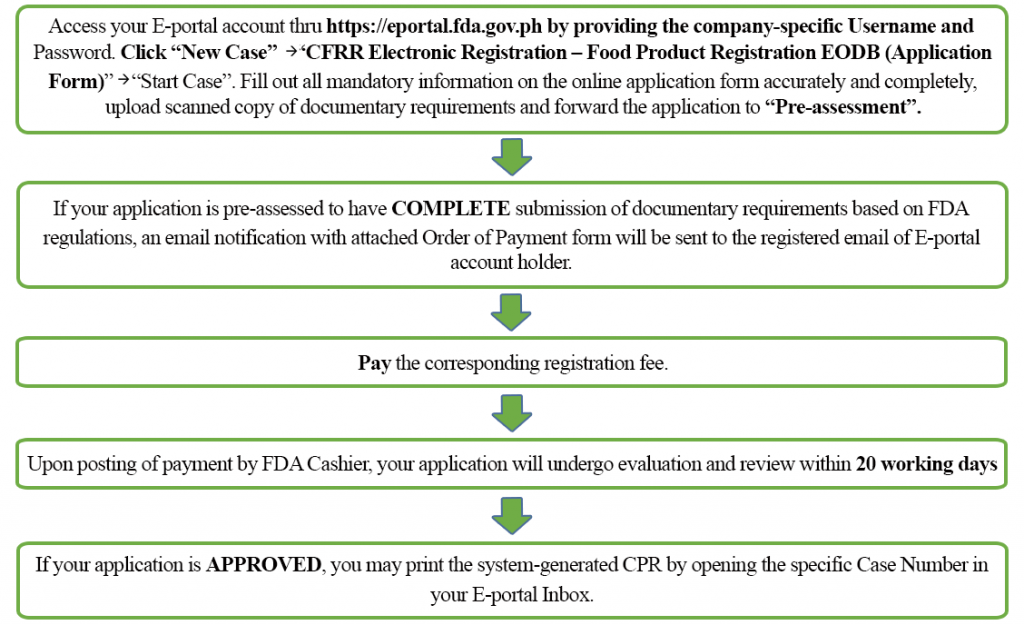
Yet, if your awarding for pre-assessment is found to have INCOMPLETE submission of documentary requirements based on FDA regulations, an email notification with fastened Results of Pre-cess will be sent to the registered e-mail of Due east-portal account holder. The Instance Number will close upon assessment of initial, subpoena, renewal or reapplication data capture applications having incomplete submission of documentary requirements. You will then need to upload again ALL previous and electric current documents and file a new application using a new case number. You lot may refer to FDA Circular No. 2020-033-A for the detailed guidelines on pre-assessment procedures in applying for CPR for nutrient in line with FDA Circular No. 2020-033.
If your successful pre-assessed application is DENIED, an email notification with fastened Letter of Denial volition be sent to the registered email of E-portal account holder. You may also download the LOD by opening the specific case number in your E-portal Inbox. File reapplication on the aforementioned case number, upload scanned copy of documents in compliance to the mentioned deficiencies and forward it to "Pre-assessment". If your reapplication is pre-assessed to accept Consummate submission of documentary requirements based on FDA regulations, an email notification with attached Order of Payment course will be sent to the registered email of E-portal account holder. Pay the corresponding reapplication fee. Otherwise, yous will receive an email notification with attached Results of Pre-assessment. Re-file the reapplication on the same example number, upload scanned copy of documents in compliance to the mentioned deficiencies and re-forward information technology to "Pre-assessment".
Y'all may likewise refer to the video on Consummate Guide on How to Apply for Food Document of Product Registration Using Eastward-portal Version 2
v. How much is the registration fee and how many years is the validity of a CPR?
Fees and charges will exist computed by the arrangement based on the number of years (2, three, 4 or 5 years validity) practical for and type of product. You lot may refer to Administrative Order No. 50-2001 for the schedule of fees. Payment may be fabricated through the FDA Cashier by presenting your printed Lodge of Payment form. Payments through Bancnet Online and LandbankOncoll are likewise accepted.
6. Do I need to submit a production sample?
Only Initial applications for Food Supplement are required to submit a representative sample in commercial presentation consistent with the Eastward-Registration application. It should exist properly labelled with the respective case number, packaged accordingly to protect the contents and submitted to the Food and Drug Administration Main Office Building within ten (10) days upon payment of assessed fee through either of the following means:
- Personal delivery to the Food and Drug Action Middle (FDAC) Unit Starmall Alabang; or
- Commitment via registered courier with the following information:
-TO: FOOD AND DRUG Assistants – Borough Drive, Filinvest City, Alabang, Muntinlupa Metropolis 1781
-FROM: Company's complete proper name and address
-Subject area: Food Production East-Registration Awarding (Case No.)
-Brand Name
-Product Proper noun
-Email address
7. Practise I need to file a separate CPR if the product volition also be exported?
A previously registered product initially for local distribution shall be allowed to be exported using the same CPR as long as the following weather are met, and labelling and standards of the importing country are as well met:
The same brand name
The aforementioned product proper name/variant
The same product formulation/ ingredients in the aforementioned gild of proportion
Subpoena application of Packaging Design for export production shall be made and labels in the language of the importing country shall as well be submitted.
ten. How practise I secure a username and password for East-Registration? Can I utilise one username and countersign for all the companies I am handling?
The CFRR E-Registration User Business relationship and Countersign is visitor-specific. An authorized representative handling multiple companies must secure a divide user account and password for each respective visitor. You must secure a notarized authority letter from the visitor (with a valid License to Operate Number) beingness represented or the company account holder. Send the asking for a User Business relationship to [e-mail protected] following the format specified beneath with the scanned notarized dominance letter of the alphabet:
Subject: CFRR:Eastward-Registration
Torso: Email Address:
Last Proper name:
First Name:
Heart Name:
Visitor Name:
The issued CFRR E-Registration User Business relationship will be sent to the due east-mail address you provided in the request. Be sure to cheque the Spam folder of your e-mail if y'all accept not received a answer within three working days.
You may too use your East-LTO User Account and Password for E-Registration by having it validated through a request sent via email to [email protected] post-obit this format:
Bailiwick: CFRR:E-Registration (Re-validate)
Trunk: Email Address:
Final Proper noun:
First Name:
Center Proper name:
Company Name:
User Account:
Countersign:
The User Account and Password validity is the same with the issued License to Operate (LTO). You volition need to renew these by sending a request via email to [email protected] following this format:
SUBJECT: CFRR: East-Registration (Renewal of User Account & Password)
Trunk: Email Address:
Last Name:
First Name:
Centre Name:
Company Proper name:
User Account:
Password:
Y'all may refer to FDA Round No. 2020-033 for other details and format of documents required in securing a User Account and Password for E-Registration.
12. How can I employ the remaining validity of the CPR of an existing production applied by other company if I volition be the new Importer?
- (From Manual Registration) this will fall under, Amendment Data Capture, Change in Importer. The new Importer must employ for CPR as amendment and must declare the current FR number, and its validity. Attach all the Initial documents stipulated in AO 2014-0029 Annex D. Delight note that the existing CPR named under the sometime Importer must be surrendered upon approving of the amendment.
- From the erstwhile E-Registration portal (version 1), this will fall under Amendment Information Capture, Change in Importer. The new Importer must apply for CPR as amendment using their own Eastward-registration account and must declare the current FR number, old example number, appointment issued and validity of CPR. Attach subpoena requirements in improver to initial requirements stipulated in AO 2014-0029 Addendum D. Please note that the existing original difficult re-create of the CPR named under the quondam Importer must exist surrendered in writing for cancelation upon approval of the subpoena.
xiii. What will be the proof that my applications under Notification of Amendment is approved or disapproved?
Applications under Notification of Amendment have no changes in CPR information (except CPR remarks, if applicable) but will yet automatically generate CPR under Generated Documents Tab. Once the case number is already in your inbox account, you lot can click the example number and view the Amendment Summary Tabular array which reflects the result of your application. If the awarding is disapproved, yous tin can see the remarks or reason for its disapproval on the right side of the tabular array.
15. What are the requirements for food supplements?
Same equally the requirements for conventional food products every bit stipulated in the Authoritative Club 2014-0029 with the improver of stability study, Certificate of Assay (COA) of the finished production and equally applicable, safety information (e.g. LD-50 Toxicity Examination). For food supplements containing herbs which are not listed in any official pharmacopeia and substances with no established safe levels.
sixteen. What level of Vitamins, Minerals and Herbs are allowed to be registered as a Food Supplement?
Vitamins and Minerals levels are computed by % RENI, every bit per Office Club 22 s.1991.To be classified as food supplement, the maximum limit is 150% RENI for Water Soluble Vitamins and 105% RENI for Fat Soluble Vitamins. For minerals, PDRI 2015 & ASEAN can exist used as reference for the maximum limit. Herbals and Botanicals that cannot be computed since information technology has no % RENI requires a justification for the rubber and test for toxicity report (LD50 Toxicity Test) and/or inclusion in the GRAS listing or Official Pharmacopeia Listing.
18. What are the information in the Shelf Life Report?
The Stability Data of the shelf life study should include: conclusion parameters used and methodology declaring the Product name, Batch number, Product date, dates of analysis, Tabulated information & results in terms of physical and chemical and Name and signature of the QA Annotator and QA Manager. We accept results for actual and accelerated shelf life study. If the shelf life study is on-going, results for at least 6 months is allowed.
LABELLING OF PREPACKAGED Food PRODUCTS
ISSUANCE OF BOC CLEARANCE
2. How practise we secure FDA Clearance (BOC Clearance) and so that the BOC tin release food products intended as donation and what are the requirements?
The requesting party should submit the hard copy of the following documents to FDAC:
- Bureau of International Wellness Cooperation (BIHC) Endorsement Letter
MARIA SOLEDAD Q. ANTONIO, Medico, PhD, MPH, DPAFP, CESE
Director 4
Agency of International Health Cooperation
- Awarding Alphabetic character or Letter of Intent– addressed to Director Pilar Marilyn M. Pagayunan, this should state the visitor's reason for importation
- Certificate of Origin or Certificate of Quality – this should have a statement on the expiration engagement of the donated products. The expiration engagement should at to the lowest degree be 6 months upon inflow in the Philippine port.
- Commercial or Proforma Invoice
- Packing Listing
- Bill of Lading or Airway Bill – submit if available, withal, the asking for clearance will still exist facilitated even though this is non submitted.
- Payment (Php 510 per Invoice)
- Letter or Document of Donation
- Deed of Credence
After the clearance is issued to the requesting party, they should contact the FDA-Regional Field Office at [email protected] for the schedule of inspection. The shipment volition not be released without inspection from FDA-RFO.
Reference: DOH Authoritative Order 2020-0001
4. Exercise we need to amend our CPR if in that location are changes in alcohol content or vintage?
No, you but have to notify the Heart for Food Regulation and Research (CFRR) on the product'southward modify in alcohol content and/or vintage.
The following documents should exist submitted:
- Awarding Letter/ Letter of the alphabet of Intent – state the changes in the wine
- Affidavit of Undertaking (Original and Notarized) – this should land that the only change in the subject wines are booze content and/or vintage
- Characterization – the submitted label should be the same with the previous label with the exception of alcohol content and/or vintage
An FDA-CFRR issued alphabetic character will be presented to BOC together with the valid CPR to facilitate the release of wines. Reference: FDA Circular 2014-022
7. How do we secure entry of products for exhibit into the Philippines?
You need to submit complete requirements as follows:
- Awarding Letter – addressed to Director Pilar Marilyn Thou. Pagayunan, this should land the company's reason for importation and the event/exhibit it volition partake in
- Affidavit of Undertaking (Original and Notarized) – a statement/clause stating that "the imported products will not be offered for auction or for commercial distribution" or similar statement that the imported products will not accomplish trade
- Document of Analysis OR Certificate of Free Sale – either of the certificate is acceptable
- Commercial or Proforma Invoice
- Packing List
- Bill of Lading or Airway Pecker – submit if available, yet, the asking for clearance will still be facilitated fifty-fifty though this is not submitted.
- Payment (Php 510 per Invoice)
SALES PROMOTION Permit Awarding
2. What are the food products for Sales Promotion covered past FDA?
All candy food and nutrient products (complete list is in AO 2014-0029) including:
Nutrient Category (Imported & Locally Manufactured)
- Bakery & Bakery Products
- Not-alcoholic beverages & beverages mixes
- Candies & confectionary products
- Cocoa & Tea and non-dairy creamer
- Condiments, sauces and seasoning
- Culinary products
- Gelatin, Desert grooming & Mixes
- Dairy Products
- Dressing & spreads
- Flour/Flour mixes & Starch
- Fish and other marine products
- Fruits, vegetable & edible fungi
- Meat and poultry products
- Noodles, pastas and pastry wrapper
- Nut and Nut products
- Native delicacies
- Oils, fats and shortenings
- Snack foods and breakfast cereal
- Sugar and other related products
- Alcoholic Beverages
- Food Supplements
- Tea (Herbal)
- Bottled Drinking H2o
- Food for Babe & Children (Growing-upwardly Milk)*
- Foods for Special Dietary Utilize
- Transgenic food products (use of genetic engineering/biotechnology)
- Ethnic food products with indigenous ingredient(s) not common in the Phil.
- Dispensed Not-Alcoholic Beverages (for discussion)
* To be referred to I.A.C–DOH
>>Marketing and practices covered by the Milk Code shall be forwarded to IAC Secretariat to be deliberated by the IAC on EO 51.
Yet, merely nutrient products duly registered with FDA are allowed to deport sales promotional activities.
3. What are exempted from the rules on Sales Promotion campaign?
- Government – sponsored Sales promotion campaigns sponsored, when the same is conducted in the practice of their governmental functions;
Individual entities in joint project/s with any authorities agency under the preceding paragraph
Social, borough, political, religious, educational, professional and other like organizations) which extend promotional activity among their members. Provided that the promotional activity is not considered sales promotion entrada every bit divers under these Rules
Reference: DAO No. two, Serial of 1993, as amended by DAO No. x-02, Series of 2010
5. What are the requirements in securing for a Sales Promo Permit?
For Initial Application
Integrated Application Form
Accomplished Data Canvas and Mechanics of the Promotion
Copy of valid CPR
Ad/ Collateral Materials to be used in the Promotion, if any
Proof of Payment
*Proof of Schedule from FDAC must besides be presented
For Subpoena
Integrated Awarding Form
Letter of Intent stating the desired changes
Re-create of canonical permit
Additional Advertising/ Collateral Materials to exist used in the Promotion, if any
Proof of Payment
Link: List of Acceptable and Not Adequate requirements for Initial and Amendment Application
6. Where are we going to submit the requirements for pre-assessment once nosotros take given the schedule?
Submit requirements to [email protected]. The application volition undergo pre-assessment based on the completeness and correctness of the requirements. Proof of Schedule from FDAC must also exist submitted. Otherwise, the submitted application will non be pre-assessed.
If the requirements are complete and compliant, an electronic mail will be sent to the company with instruction to go along with the payment.
If the application is incomplete, the bidder will be advised to secure a new appointment schedule to submit the application for pre-cess. The client must request for a new DTN and schedule of engagement.
*A pre-cess grade will exist attached on the email.
8. How much is the payment?
The fee depends on the coverage of the promotional activity and amount of prizes to be won (per DTI-DOH Joint AO No. one s. 2000)
| Coverage | Fee |
| NCR only or in several regions in NCR and Nationwide | Php 1, 010 |
| More than one (one) region but excluding NCR | Php 760 |
| Several provinces/ cities/ municipalities within a single region | Php 560 |
| Single province/ urban center/ municipality | Php 260 |
| Amount of Prizes | Fee |
| 150,000.00 & below - 300,000.00 | Php 1,010.00 |
| 300,001.00 - 500,000.00 | Php 2,020.00 |
| 500,001.00 - 1,000,000.00 | Php 3,030.00 |
| Above 1, 000,000.00 | Php 5,050.00 |
For amendment including extension, the fee is Php 310.00, merely depends if the amount of prizes will be increased.
9. To which Center should we file our application if the sales promotion volition cover combined products of different categories?
| Products Involved | Concerned Center |
| Drugs, Nutrient, Cosmetics, Device, HHS (or Drug with whatsoever categories) | Eye for Drug Regulation and Enquiry (CDRR) |
| Food, Cosmetics, Device, HHS (or Device with any product categories excluding Drug) | Center for Device Regulation, Radiation Health and Research (CDRRHR) |
| Food, Cosmetics, HHS | Center for Food Regulation and Research (CFRR) |
| Cosmetics and HHS | Centre for Cosmetic Regulation and Research |
13. What are the General Rules on Advertisements, Promotions, Sponsorship and other Marketing Activities of any health products?
As per Volume II, Commodity 5 of IRR of RA 9711
- No health product that has not been registered or authorized shall exist advertised, promoted or subjected to whatsoever marketing activities;
- No merits in the advertisement, promotion and sponsorship, and other marketing activities shall be made other than those contained in the approved characterization or packaging of the wellness product, or every bit duly canonical by the FDA;
- No claims, therapeutic or scientific otherwise, shall be fabricated that has not been duly canonical by the FDA;
- All health products that are permitted to exist promoted must specifically land the authority or reference number that approved the same promotional, sponsorship, or marketing activities.
LIST OF Adequate AND NOT ACCEPTABLE REQUIREMENTS FOR SALES PROMO Let INITIAL AND Subpoena APPLICATION
INITIAL Application
| INITIAL APPLICATION | |||
| REQUIREMENTS | ACCEPTABLE | Not ACCEPTABLE | BASIS |
| ane.Integrated Application Form | -Check the consistency of applicant's name, establishment's name and address indicated in the Integrated Application Form -Check the applicant's signature | -Non-submission -Submitted integrated application form contains incomplete information | FDA Circular No. 2014-003 |
| 2. Information Sail and Mechanics of Sales Promotion | -It contains iii sheets and all must be properly filled out -Promo title -Elapsing -Coverage -Promo Mechanics | -Non-submission -Submitted information sheet and mechanics of sales promotion contains incomplete information -Practical promo duration has already started or lapsed -With Drug and/or Device product -Duration is across three months for discount scheme and 6 months for closing out sales -No coverage included -With products covered by the Milk Code or EO 51 | Republic Deed 7394 Executive Club No. 51 or the Milk Code FDA Memorandum Circular No. 2013-028 DTI-DOH Joint Authoritative Lodge No. 01 serial of 2000 Codex Stan 180-1991 |
| 3. Duly and validly registered participating products | -Valid Certificate of Product Registration for Food products -Valid Cosmetic Notification for Corrective products* *In example there are applications with combined food and cosmetic products | -Expired Certificate of Product Registration for Nutrient products -Expired Cosmetic Notification for Corrective products* (products undergoing renewal non allowed) | Republic Deed 7394 |
| four. Promotional Materials, if whatever | -Articulate and readable promotional materials -The line "DOH-FDA-CFRR-Allow No. xxxx s. xxxx must be indicated | -Not readable promotional materials - The line "DOH-FDA-CFRR-Let No. xxxx southward. xxxx is not indicated. -With obvious health and nutritional claims and therapeutic claims and/or claims that are non included in the approved label. e.g. Can cure diabetes Can lower blood cholesterol Superior corporeality of Vitamin C -With sexy tones, skin exposure, potentially controversial/sensitive execution -Employ of the FDA Logo and Name - Non-inclusion of the Filipino Standard Message in prescribed format: "Mahalagang Paalala: Ang (proper name of product) ay hindi gamot at hindi dapat gamitin panggamot sa anumang uri ng sakit" for sales promotion application with Food Supplements equally participating products | Republic Act 7394 Agency Circular No. 2007-002 FDA Memorandum Circular No. 2013-030 Administrative Order 2010-0008 |
| five. Proof of Payment of Fees | -Payment details must be posted and verified by the FDA Cashier in the IFS-Document Tracking Organisation | -Non-submission - Payment details are not posted and verified past the FDA Cashier | |
AMENDMENT APPLICATION
| Amendment APPLICATION | |||
| REQUIREMENTS | FOR APPROVAL | GROUNDS FOR DENIAL | Ground |
| 1.Integrated Application Course | -Check the consistency of applicant's name, establishment's proper noun and address indicated in the Integrated Awarding Form -Check the applicant'due south signature | -Not-submission -Submitted integrated application grade contains incomplete information | FDA Circular No. 2014-003 |
| 2. Letter of the alphabet of Intent stating the specific changes | -It should country the specific changes made from the previously approved to proposed modify/s. | It does non clearly state specific modify/southward made. Made change/due south not included in the letter of intent. | Republic Act 7394 |
| 3. Copy of initially approved Sales Promo Permit | -Clear and readable -Should include all annexes attached | -Non readable | Administrative Order 4-A s, 1995 |
| iv. Promotional Materials, if whatever | -Clear and readable promotional materials -The line "DOH-FDA-CFRR-Permit No. xxxx south. xxxx must exist indicated | -Not readable promotional materials - The line "DOH-FDA-CFRR-Permit No. xxxx s. xxxx is not indicated. -With obvious health and nutritional claims and therapeutic claims e.one thousand. Can cure diabetes Can lower claret cholesterol Superior corporeality of Vitamin C -With sexy tones, skin exposure, potentially controversial/sensitive execution -Utilise of the FDA Logo and Proper name - Non-inclusion of the Filipino Standard Message in prescribed format: "Mahalagang Paalala: Ang (name of production) ay hindi gamot at hindi dapat gamitin panggamot sa anumang uri ng sakit" for sales promotion application with Food Supplements as participating products | Democracy Act 7394 Bureau Circular No. 2007-002 FDA Memorandum Circular No. 2013-030 Administrative Order 2010-0008 |
| 5. Proof of Payment of Fees | -Payment details must be posted and verified by the FDA Cashier in the IFS-Certificate Tracking System | -Not-submission - Payment details are non posted and verified by the FDA Cashier | |
FOOD SAFETY UNIT
POLICY Research AND STANDARDS Development Division
1. What are the sweetened beverages (SB) field of study to excise tax under Section 47 of the Democracy ACT 10963 (Train LAW)?
The SBs subject to excise tax are not-alcoholic beverages in liquid, powder or concentrates that are pre-packaged and sealed in accordance with FDA standards, that contain caloric sweetener and/or not-caloric sweeteners added by the manufacturers, and shall include, but not express to the following, as described in the Nutrient Category System from Codex Stan 192-1995 every bit adopted by the FDA:
- a) Sweetened juice drinks;
- b) Sweetened tea;
- c) All carbonated beverages;
- d) Flavored water;
- e) Free energy and sports drinks;
- f) Other powdered drinks not classified every bit milk, juice, tea and coffee;
- g) Cereal and grain beverages; and
- h) Other non-alcoholic beverages that contain added sugar.
two. What are the beverages exempted from SB excise tax?
Section 47 (A) Rate and Base of Revenue enhancement (1) states that sweetened beverages using purely coconut sap sugar and purely steviol glycosides shall be exempt from tax.
Moreover, Department 47 (C) Exclusions indicates that the following beverages are exempted from taxation:
- All milk products including plain milk, infant formula milk, follow-on milk, growing upwards milk, powdered milk, ready-to-drink milk and flavored milk, fermented milk, soymilk, and flavored soymilk;
- One hundred pct (100%) natural fruit juices – Original liquid resulting from the pressing of fruit, the liquid resulting from the reconstitution of natural fruit juice concentrate, or the liquid resulting from the restoration of water to dehydrated natural fruit juice that practice non accept added carbohydrate or caloric sweetener;
- One hundred percent (100%) natural vegetable juices – Original liquid resulting from the pressing of vegetables, the liquid resulting from the reconstitution of natural vegetable juice concentrate, or the liquid resulting from the restoration of water to dehydrated natural vegetable juice that exercise not have added saccharide or caloric sweetener;
- Repast replacement and medically indicated beverages – Any liquid or powder drink/product for oral nutritional therapy for persons who cannot absorb or metabolize dietary nutrients from food or beverages, or as a source of necessary nutrition used due to a medical status and an oral electrolyte solution for infants and children formulated to prevent dehydration due to illness; and
Basis coffee, instant soluble java and prepackaged powdered coffee products.
3. What are caloric sweeteners?
According to Train Law, caloric sweeteners are substances that are sweet and includes sucrose, fructose, and glucose that produces a certain sweetness.
4. What are non- caloric sweeteners?
According to TRAIN Police force, non- caloric sweeteners are substances that are artificially or chemically processed that produces a sure sugariness. These substances which tin can be directly added to beverages include aspartame, sucralose, saccharin, acesulfame potassium, neotame, cyclamates, and other not-nutritive sweeteners approved by the Codex Alimentarius and adopted by the FDA.
five. What is high fructose corn syrup (HFCS)?
Co-ordinate to Railroad train Constabulary, HFCS refers to a sugariness saccharide mixture containing fructose and glucose which is derived from corn and added to provide sweet to beverages, and which includes other similar syrup preparations.
half dozen. What is kokosnoot sap sugar?
Co-ordinate to PNS/BAFPS 76:2010, coconut sap carbohydrate is a nutritive sweetener in solid grade containing sucrose, glucose, and fructose, and derived from pure fresh kokosnoot sap obtained by boiling.
vii. What is steviol glycosides?
According to FAO JECFA Monograph 23 (2019), steviol glycosides is a mixture of compounds extracted from the leaves of Stevia rebaudiana Bertoni containing a steviol backbone conjugated to any number or combination of the principal carbohydrate moieties (glucose, rhamnose, xylose, fructose, arabinose, galactose and deoxyglucose) in any of the orientations occurring in the leaves of Stevia rebaudiana Bertoni. It is a white to light yellow powder, odourless or having slight characteristic odour, and well-nigh 200 to 300 times sweeter than sucrose.
nine. Does the FDA issue SB excise tax exemption certificate?
No. The FDA does not issue tax exemption certificate. It only provides responses to letter requests, stating the production categorization/classification according to Codex Stan 192-1995 and/or blazon of sweeteners used in the drinkable product. The Agency of Internal Revenue (BIR) or Bureau of Customs (BOC) determines whether a production is tax exempt or not, and/or how much taxation is due based on the information on production classification co-ordinate to Codex and/or blazon of sweeteners used provided by FDA.
Center for Cosmetics and Household Urban and Chancy Substances Regulation and Research
Eye for Device Regulation, Radiation, Wellness and Research
Do I Need To Register With Fda To Get My Label Approved,
Source: https://www.fda.gov.ph/faqs/
Posted by: fontanaalmyconver.blogspot.com


0 Response to "Do I Need To Register With Fda To Get My Label Approved"
Post a Comment